- Blog
- About
- Contact
- Whitney houston grammys one moment in time
- Kanaa movie tamilrockers
- Realtek 11n usb wireless lan utility amazon
- Garmin inreach login
- Destroy all humans path of the furon ps3 gamestop
- Kanto karaoke torrent download
- Free dreamweaver templates with flash
- Castle miner z pc game
- India arie worthy tour chicago
- 3d movies for oculus quest
- Antique tavannes pocket watch
- Gameplay of multiwinia
- How to calculate ppm from mole fraction
- Midico mac karaoke
- Qing fei de yi lee min ho mp3
- Maschine library logic pro x
- What is stardust
- Examples of weather symbols in the great gatsby
- Different pc fan sizes
- Stencyl backgrounds
- Scan new plugins in protools 12
- Play digging jim online free
- Sprained acl
- Price ultimate guitar pro
- Driver printer zebra lp 2844 z
- Steve jobs 2015
Worked Examples: Converting Percentage Concentration to ppmįollow these 4 steps to convert percentage concentration to parts. M/v (w/v) and m/m (w/w) concentrations and parts per million will be discussed in the following section (after the worked examples of percentage concentration and ppm below).

V/v concentration is NOT the same as a %(v/v) concentration M/m concentration is NOT the same as a %(m/m) concentration W/v concentration is NOT the same as a %(w/v) concentration M/v concentration is NOT the same as a %(m/v) concentration Where x is the value of the percentage concentration This is true for other similar percentage concentrations:įor mass/volume percentage concentrations (m/v% or w/v%):įor mass/mass percentage concentrations (m/m% or w/w%): Calculating solution concentrations using Molarity. We could write a mathematical expression equating parts per hundred (a percentage) and parts per million (ppm) as shown below: Molarity is defined as the moles of a solute per volume of total solution. How would we convert that to a concentration in parts per million (ppm) ? Formula: concentration (mg/m 3) 0.0409 x concentration (ppm) x molecular weight For example: 100 ppm of NH 3 (17.03 g/mol) 0.0409 x 100 ppm x 17.03 69. This means that there are 9 000 parts of NaCl in every 1 000 000 parts of the solution.īut the concentration of a solution is sometimes given as a percentage.Ī percentage concentration tells you how many parts of solute are present in 100 parts of solution.įor example, the ethanol content in wine is often given as about 12%(v/v), that is, 12% of the volume of the wine is ethanol, or, there are 12 parts of ethanol in every 100 parts of solution. The temperature of the atmosphere also has an influence on the calculation. Recall that, in general, concentration tells you how much solute is present in a solution.Ī concentration in parts per million (ppm) tells you how many parts of solute are present in 1 000 000 parts of solution.įor example, a saline solution is a dilute aqueous solution of sodium chloride, NaCl (aq), with a concentration of 9 000 ppm. Jan 2008 - 38 What is the concentration of O 2(g), in parts per million, in a solution that contains 0.008 gram of O 2(g) dissolved in 1000.Parts per million and Percentage Concentration Calculations grams of a solution having a concentration of 5 parts per million? June 2009- 36 What is the total mass of solute in 1000. June 2008- 38 What is the concentration of O 2(g), in parts per million, in a solution that contains 0.008 gram of O 2(g) dissolved in 1000. June 2005 -42 What is the concentration of a solution, in parts per million, if 0.02 gram of Na 3PO 4 is dissolved in 1000 grams of water? O 2 Ne H 2 S Ar Total Moles 18.00 Mole fraction 1 Pressure fraction 1 Partial Pressure 1620.0 : 70.

grams of H 2O, what is the concentration of the resulting solution, in parts per million? A tank contains 5.00 moles of O 2, 3.00 moles of neon, 6.00 moles of H 2 S, and 4.00 moles of argon at a total pressure of 1620.0 mm Hg. If 0.025 gram of Pb(NO 3) 2 is dissolved in 100. Here was another one, but it was a multiple choice. It is technically wrong not to add the solute and solvents together to determine the mass of solution. Even though mathematically it didn't change a thing. Īnswer-The equation above reads "grams of solution", you had to add the solute to the solvent in the denominator. Your response must include both a correct numerical setup and the calculated result. Calculate the concentration of NaCl in parts per million (ppm). In the space in your answer booklet, calculate the dissolved oxygen concentration of this solution in parts per million. 150mL of an aqueous sodium chloride solution contains 0.0045g NaCl. An aqueous solution has 0.0070 gram of oxygen dissolved in 1000. Most students lost 1 of the 2 points on this one.Ħ6.
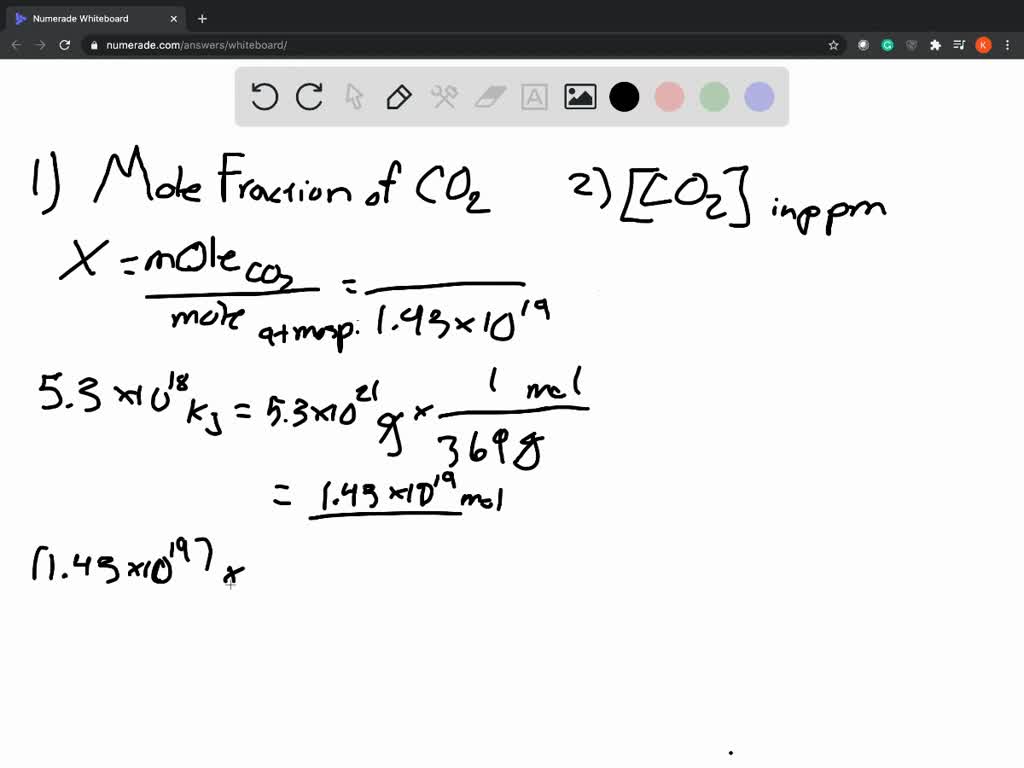
Then June 2007, New York State threw this curveball. What is the concentration of a solution, in parts per million, if 0.02 gram of NaCl is dissolved in 1000. This is really just a plug and chug problem. Think about this 1ppm = 1 inch in 16 miles This is used for very small concentrations of particles in solutions. This is another way of determining concentration.
